|
7/27/2023 0 Comments Generic name for baytril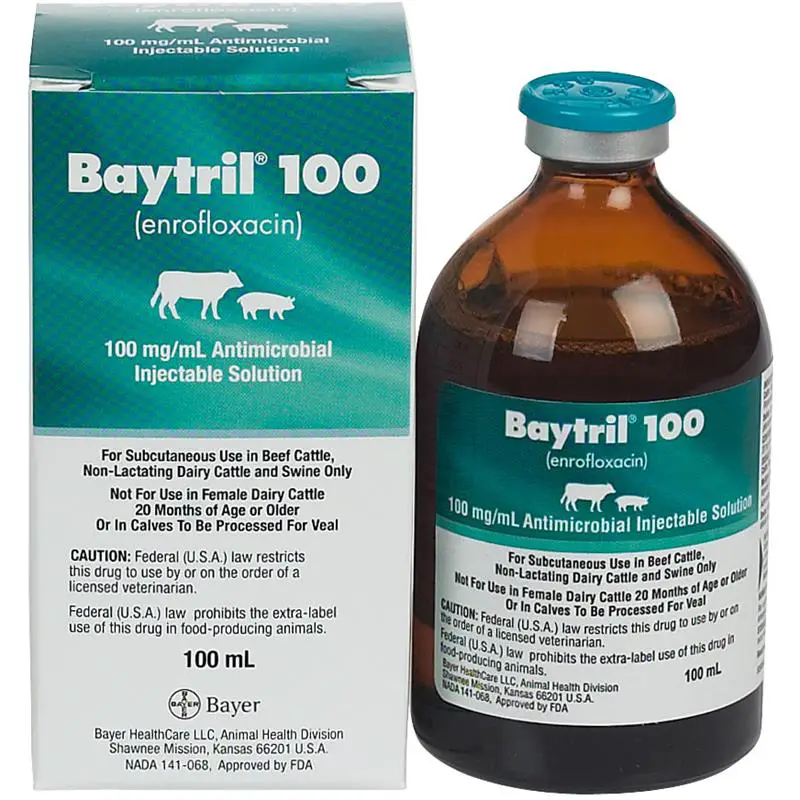
§ 530.41(a)(10)), and, in particular, extralabel use of enrofloxacin in food-producing animals ( 21 C.F.R. ” FDA’s regulations prohibit the extralabel use of fluoroquinolones in food-producing animals ( 21 C.F.R. FDC Act § 512(c)(2)(A)(ii), as amended by GADPTRA, states that FDA will approve an ANADA unless, among other things, the Agency finds that “the conditions of use prescribed, recommended, or suggested in the proposed labeling are not reasonably certain to be followed in practice. 5,756,506 (“the ‘506 patent”), which expires on June 27, 2015, and claims the use of enrofloxacin in a single high dose to replace multiple lower doses in bovine respiratory disease and swine pneumonia. 
The Single-Dose Therapy use is covered by U.S. Baytril® 100 is approved for two different dosing regimens: (1) Single-Dose Therapy (7.5 to 12.5 mg/kg of body weight (3.4 to 5.7 mL/100 lb)) and (2) Multiple-Day Therapy (2.5 to 5.0 mg/kg of body weight (1.1 to 2.3 mL/100 lb)). 1585 (1988), the Hatch-Waxman equivalent in the animal drug world – and served as a constructive denial of Bayer’s 2006 Citizen Petition in violation of the APA. 200-495 for use in cattle violates the Administrative Procedure Act (“APA”) and FDC Act § 512(c)(2)(A)(ii) – as added by the Generic Animal Drug and Patent Term Restoration Act of 1988 (“GADPTRA”), Pub. That stay was memorialized in a Stipulated Stay and Preliminary Injunction agreed to by the court, but opposed by Norbrook, and cited in an ApOrder remanding the case to FDA for reconsideration.Īs we previously reported, Bayer sued FDA on April 10th alleging that the Agency’s approval of ANADA No. District Court for the District of Columbia suspending the ANADA approval (and its associated labeling) for use in cattle (but not in swine), administratively suspended the cattle approval pending completion of the Agency’s consideration of issues raised by Bayer in a June 2006 Citizen Petition (Docket No. (“Norbrook”) for Enroflox™ 100, a generic version of Bayer’s fluroquinolone animal drug Baytril® 100 (enrofloxacin) Injectable Solution, has paid off. 200-495 submitted by Norbrook Laboratories, Ltd. Dermal infections: Enrofloxacin concentrates in inflamed tissue providing high drug levels at the infection site.Bayer HealthCare, LLC’s (“Bayer’s”) court challenge to FDA’s approval of Abbreviated New Animal Drug Application (“ANADA”) No.Achieves therapeutic concentrations in pulmonary and bronchial tissues. Respiratory tract infections: Enrofloxacin irreversibly inhibits a bacterial enzyme resulting in rapid bactericidal action. 
Provides effective management of acute or chronic infections of the genitourinary tract and prostate. Urinary tract infections: Enrofloxacin exerts rapid bactericidal action in the urinary tract.Putney Enrofloxacin Flavored Tablets delivers the same efficacy and safety profile as Baytril, but is more affordable. Enrofloxacin is bactericidal against a wide range of gram positive and gram negative bacteria by interfering with bacterial DNA metabolism killing the bacteria. Enrofloxacin Flavored Tablets (generic of Baytril) is a fluroquinolone antibiotic indicated for the management of disease in dogs and cats associated with bacteria susceptible to enrofloxacin.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
